Table 1 from he oxygen reduction reaction mechanism on Pt ( 111 ) from density functional heory calculations | Semantic Scholar
![PDF] New Pd-Pt and Pd-Au catalysts for an efficient synthesis of H2O2 from H2 and O2 under very mild conditions | Semantic Scholar PDF] New Pd-Pt and Pd-Au catalysts for an efficient synthesis of H2O2 from H2 and O2 under very mild conditions | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b6b16d7b080f74bbb2b2b4709f7613f825e59bab/2-Figure1-1.png)
PDF] New Pd-Pt and Pd-Au catalysts for an efficient synthesis of H2O2 from H2 and O2 under very mild conditions | Semantic Scholar

Dissociative adsorption of O2 on strained Pt(111) - Physical Chemistry Chemical Physics (RSC Publishing)
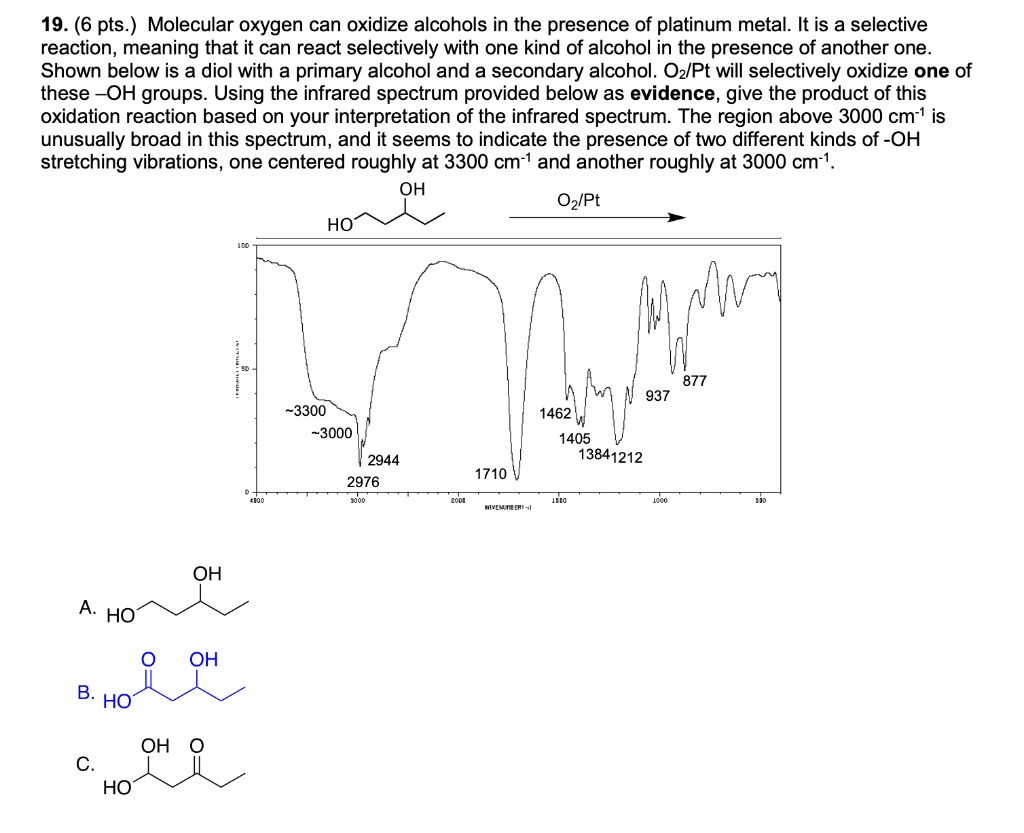
SOLVED: 19. (6 pts Molecular oxygen can oxidize alcohols in the presence of platinum metal. It is a selective reaction, meaning that it can react selectively with one kind of alcohol in
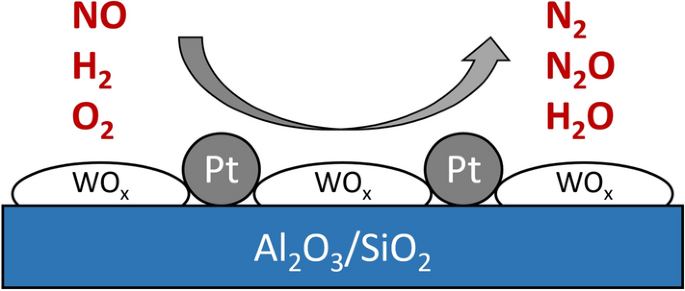
Reduction of NOx by H2 on WOx-Promoted Pt/Al2O3/SiO2 Catalysts Under O2-Rich Conditions | SpringerLink

a) H2‐TPR and b) O2‐TPD patterns of TiO2, PT, AT, and PAT samples. c)... | Download Scientific Diagram

Theoretical insights into the activation of O2 by Pt single atom and Pt4 nanocluster on functionalized graphene support: Critical role of Pt positive polarized charges - ScienceDirect

H2O-assisted O2 reduction by H2 on Pt and PtAu bimetallic nanoparticles: Influences of composition and reactant coverages on kinetic regimes, rates, and selectivities - ScienceDirect