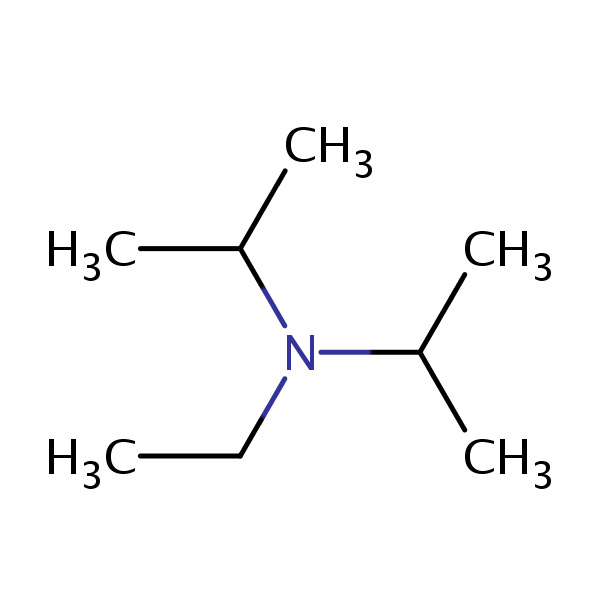
DIPEA (N,N-diisopropylethylamine, Hunig's base) molecule. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles: hydrogen (hidden), carbon (grey Stock Photo - Alamy

A Homage to Siegfried Hünig and His Research - Reissig - 2021 - Angewandte Chemie International Edition - Wiley Online Library
![PDF] An efficient and operationally convenient general synthesis of tertiary amines by direct alkylation of secondary amines with alkyl halides in the presence of Huenig's base | Semantic Scholar PDF] An efficient and operationally convenient general synthesis of tertiary amines by direct alkylation of secondary amines with alkyl halides in the presence of Huenig's base | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/37998da1cde9c148c08633c2616b218cf5a07970/4-Table1-1.png)
PDF] An efficient and operationally convenient general synthesis of tertiary amines by direct alkylation of secondary amines with alkyl halides in the presence of Huenig's base | Semantic Scholar
![Selective Syntheses of Bis[1,2]dithiolo[1,4]thiazines and Bis[1,2]dithiolopyrroles from Hünig's Base | The Journal of Organic Chemistry Selective Syntheses of Bis[1,2]dithiolo[1,4]thiazines and Bis[1,2]dithiolopyrroles from Hünig's Base | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/jo971864e/asset/images/large/jo971864en00001.jpeg)
Selective Syntheses of Bis[1,2]dithiolo[1,4]thiazines and Bis[1,2]dithiolopyrroles from Hünig's Base | The Journal of Organic Chemistry
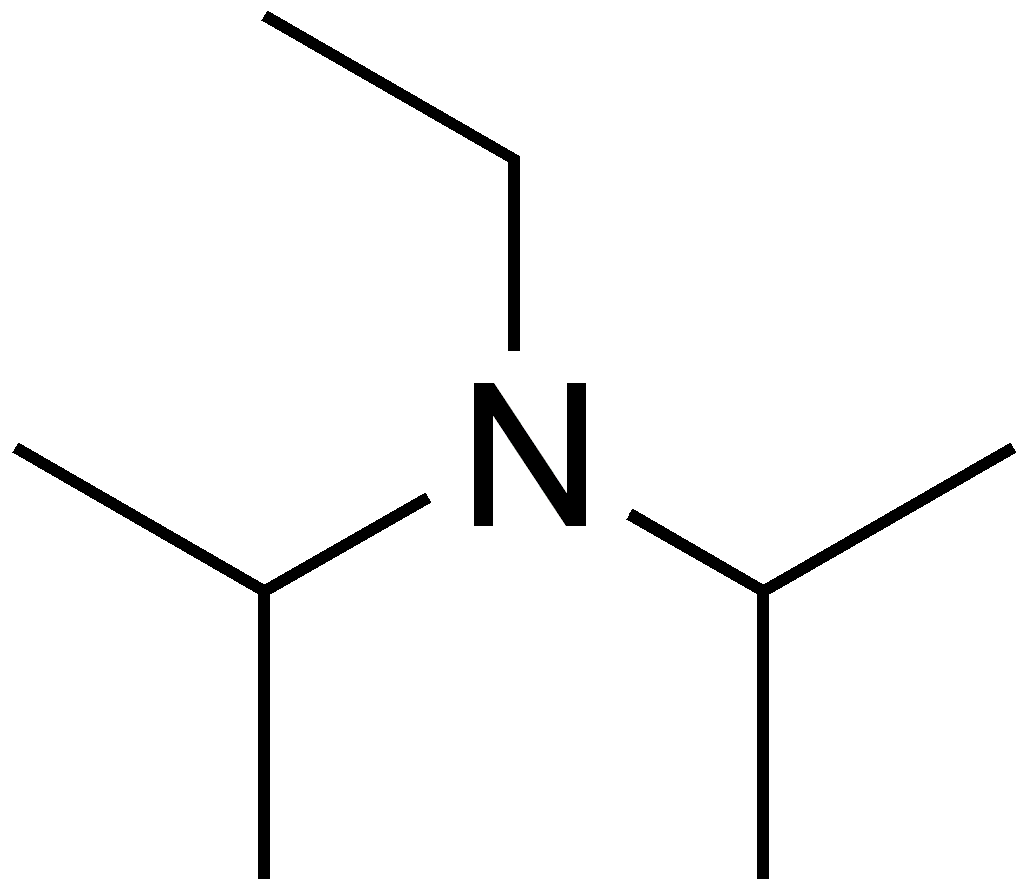
DIPEA N,N-diisopropylethylamine, Hunig`s Base Molecule. Skeletal Formula. Stock Illustration - Illustration of structure, base: 187972769

Dipea Nndiisopropylethylamine Hunigs Base Molecule Skeletal Stock Vector (Royalty Free) 1093026992 | Shutterstock
![PDF] A SIMPLE AND HIGHLY EFFICIENT SYNTHESIS OF QUINOLINE TERTIARY AMINES CATALYZED BY HUNIG'S BASE | Semantic Scholar PDF] A SIMPLE AND HIGHLY EFFICIENT SYNTHESIS OF QUINOLINE TERTIARY AMINES CATALYZED BY HUNIG'S BASE | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3c974f939a32262aeed197ab08f1ab631c012982/4-Table2-1.png)