
Stable positive electrolyte containing high-concentration Fe2(SO4)3 for vanadium flow battery at 50 °C - ScienceDirect

Ferric Sulfate Granular Fe2(so4)3 Manufacturer - Buy Ferric Sulfate Granular,Ferric Sulfate,Ferric Sulfate Manufacturer Product on Alibaba.com

How to Balance Fe + H2SO4 = FeSO4 + Fe2(SO4)3 + H2O + SO2 (Iron + Concentrated Sulfuric acid) - YouTube




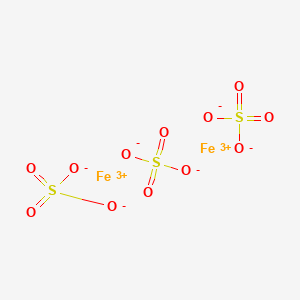








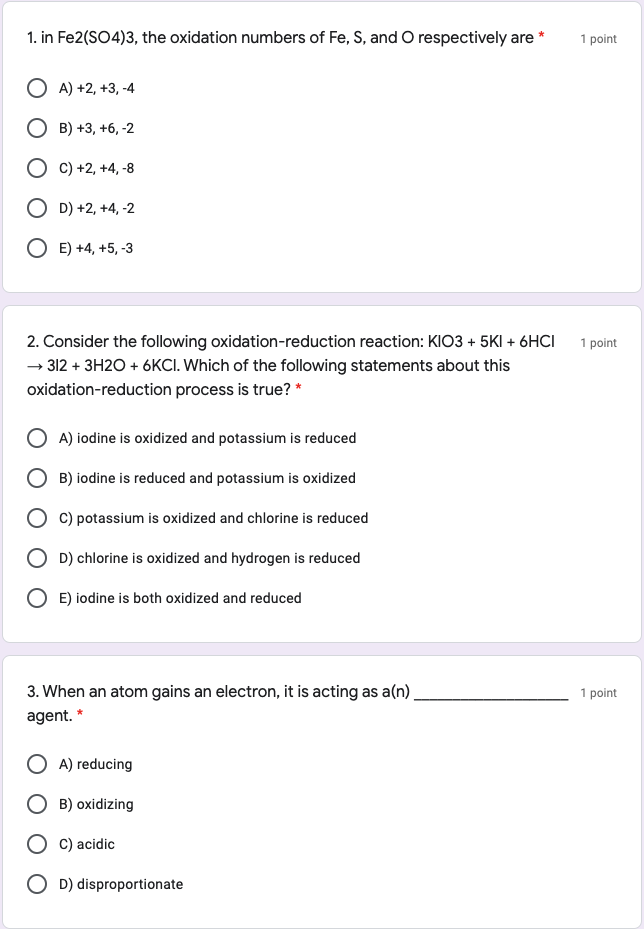

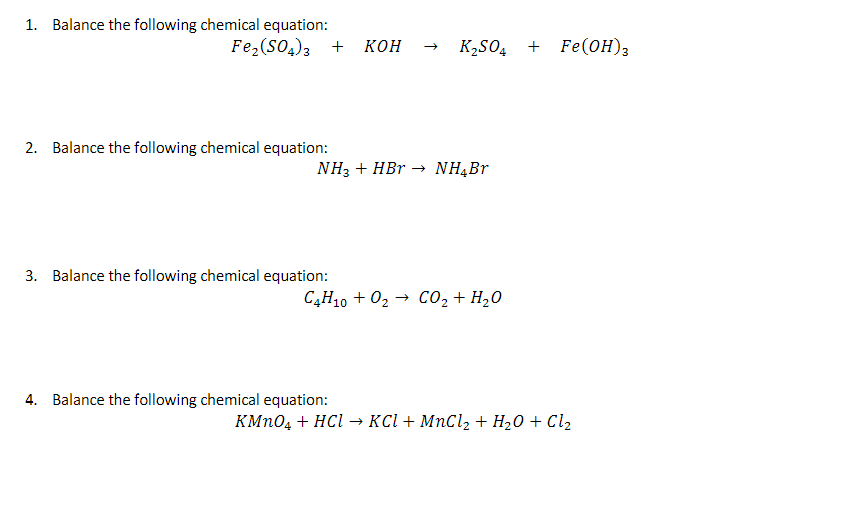

![PDF] Fe2(SO4)3·H2SO4·28H2O, a low-temperature water-rich iron(III) sulfate. | Semantic Scholar PDF] Fe2(SO4)3·H2SO4·28H2O, a low-temperature water-rich iron(III) sulfate. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3a1ee865d6b22d384850851082185de0c92f6121/1-Figure1-1.png)