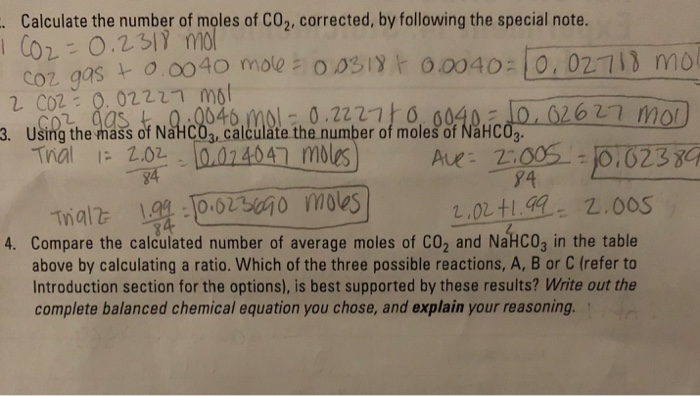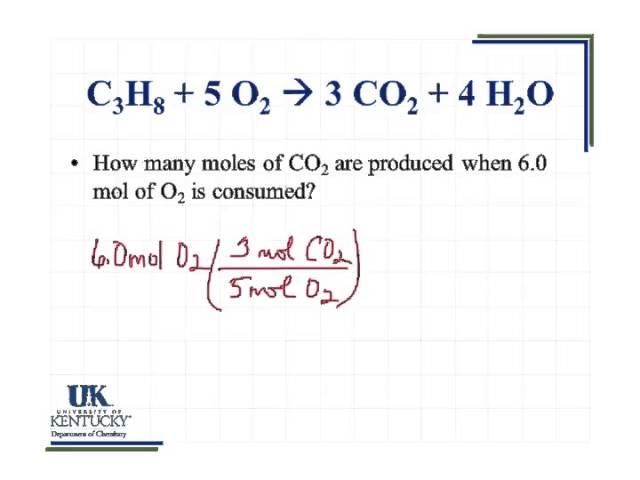Convert 22 g of carbon dioxide `(CO_(2))` into moles. (Atomic masses : `C = 12 u, O = 16 u`) - YouTube

Volume of titration, number of mol CO2, and weight of C for every KOH... | Download Scientific Diagram

How many moles of CO2 are produced when 2.0 moles of C3H8 react with an excess of O2? Remember, the products of this reaction are CO2 and H2O. - YouTube