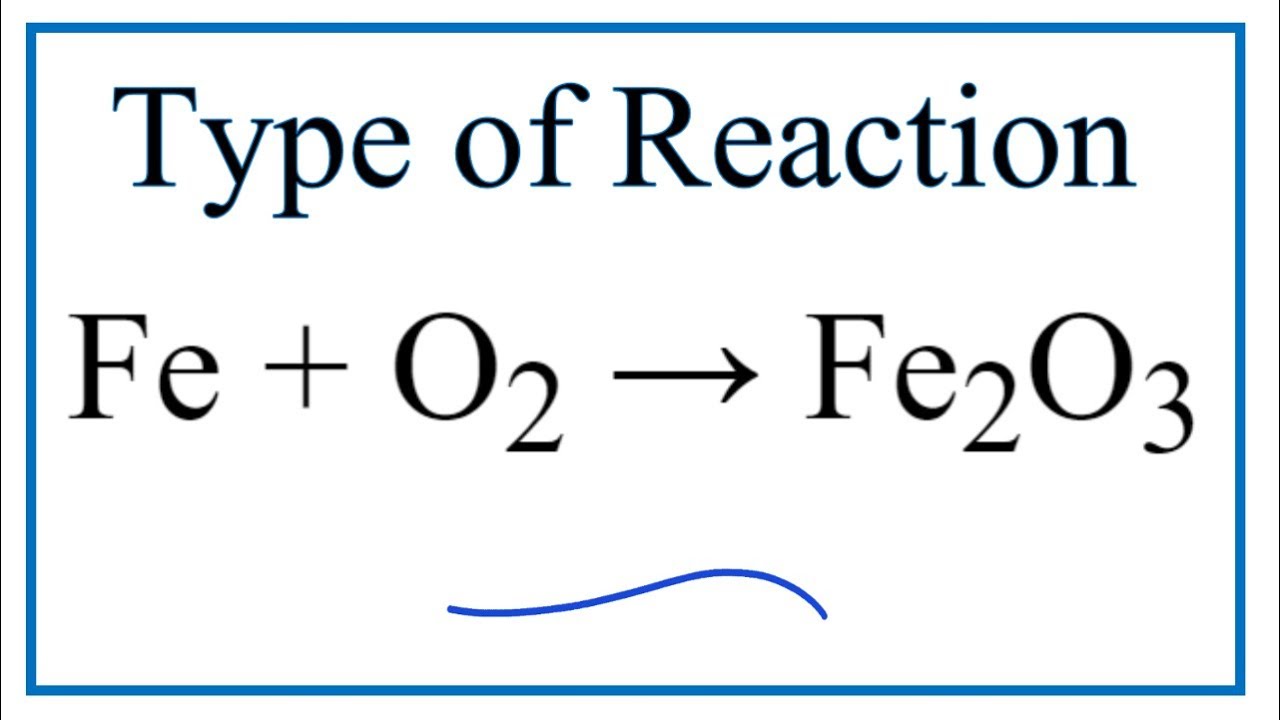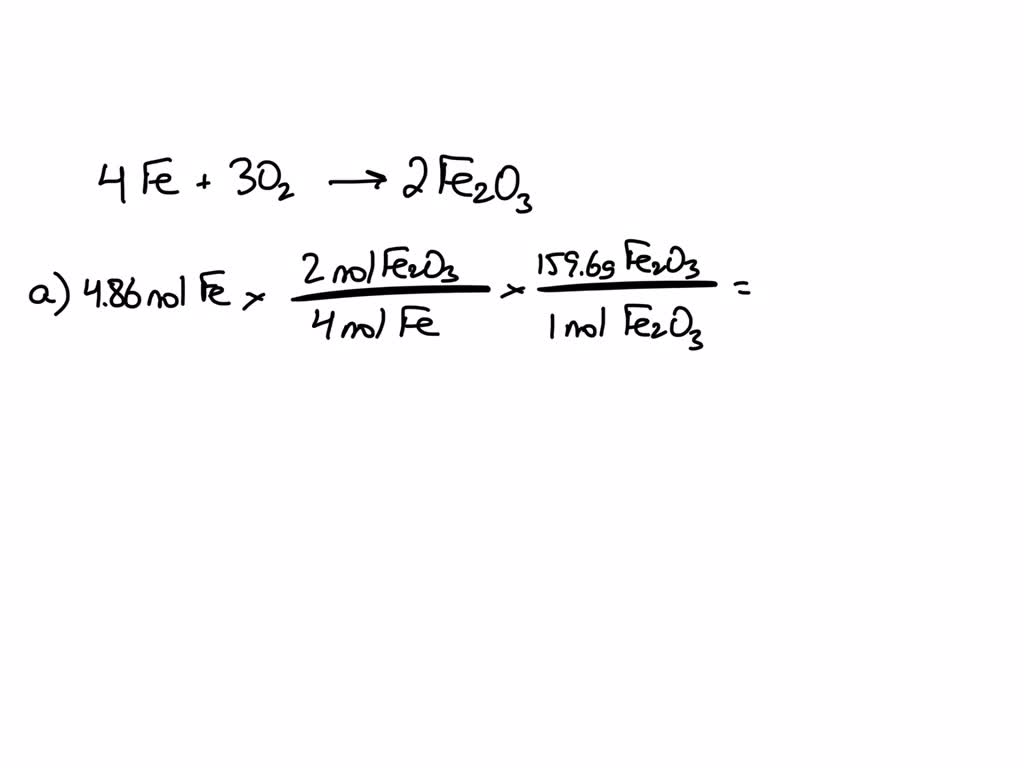
SOLVED: Given the reaction 4Fe + 3O2= 2Fe2O3 a. How many grams of Fe2O3 will be formed from 4.86 moles Fe reacting with sufficient oxygen gas? b. How many grams of Fe

SOLVED: For the oxidation of iron to form iron(III) oxide: 4Fe(s) + 3O2(g) ⇌ 2Fe2O3(s) ΔS° = –549.73 J/K at 298 K The enthalpy of formation of Fe2O3(s) is –824.2 kJ/mol. What